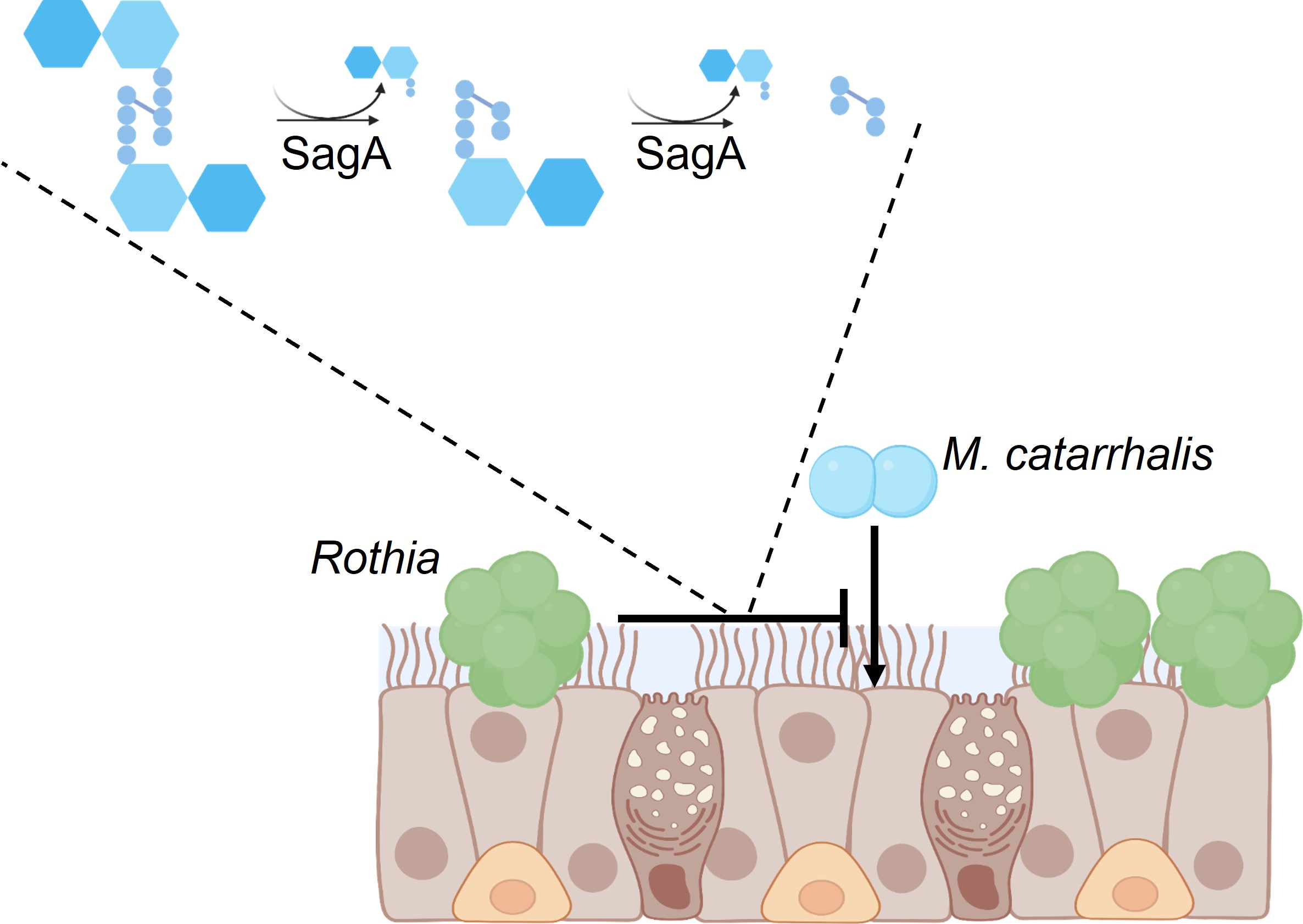

Moraxella catarrhalis is a pathobiont of the respiratory tract, responsible for ear infections in children and wheezing illnesses in children and adults with chronic respiratory diseases. Detection of M. catarrhalis during wheezing episodes in early life is associated with the development of persistent asthma. Moraxella catarrhalis is found almost exclusively within the human respiratory tract. This pathobiont is associated with ear infections and the development of respiratory illnesses, including allergies and asthma. Given the limited ecological distribution of M. catarrhalis, we hypothesized that we could leverage the nasal microbiomes of healthy children without M. catarrhalis to identify bacteria that may represent potential sources of therapeutics. Rothia was more abundant in the noses of healthy children compared to children with cold symptoms and M. catarrhalis. We cultured Rothia from nasal samples and determined that most isolates of Rothia dentocariosa and “Rothia similmucilaginosa” were able to fully inhibit the growth of M. catarrhalis in vitro, whereas isolates of Rothia aeria varied in their ability to inhibit M. catarrhalis. Using comparative genomics and proteomics, we identified a putative peptidoglycan hydrolase called secreted antigen A (SagA). This protein was present at higher relative abundance in the secreted proteomes of R. dentocariosa and R. similmucilaginosa than in those from non-inhibitory R. aeria, suggesting that it may be involved in M. catarrhalis inhibition. We produced SagA from R. similmucilaginosa in Escherichia coli and confirmed its ability to degrade M. catarrhalis peptidoglycan and inhibit its growth. We then demonstrated that R. aeria and R. similmucilaginosa reduced M. catarrhalis levels in an air-liquid interface culture model of the respiratory epithelium. Together, our results suggest that Rothia restricts M. catarrhalis colonization of the human respiratory tract in vivo. Highlighted in mBio and Nature Microbiology.